Research Ethics Committee
The primary purpose of the JMCFI-Research Ethics Committee is to safeguard the rights, well-being, and dignity of research participants while promoting the highest standards of research integrity.
Research Ethics
Committee
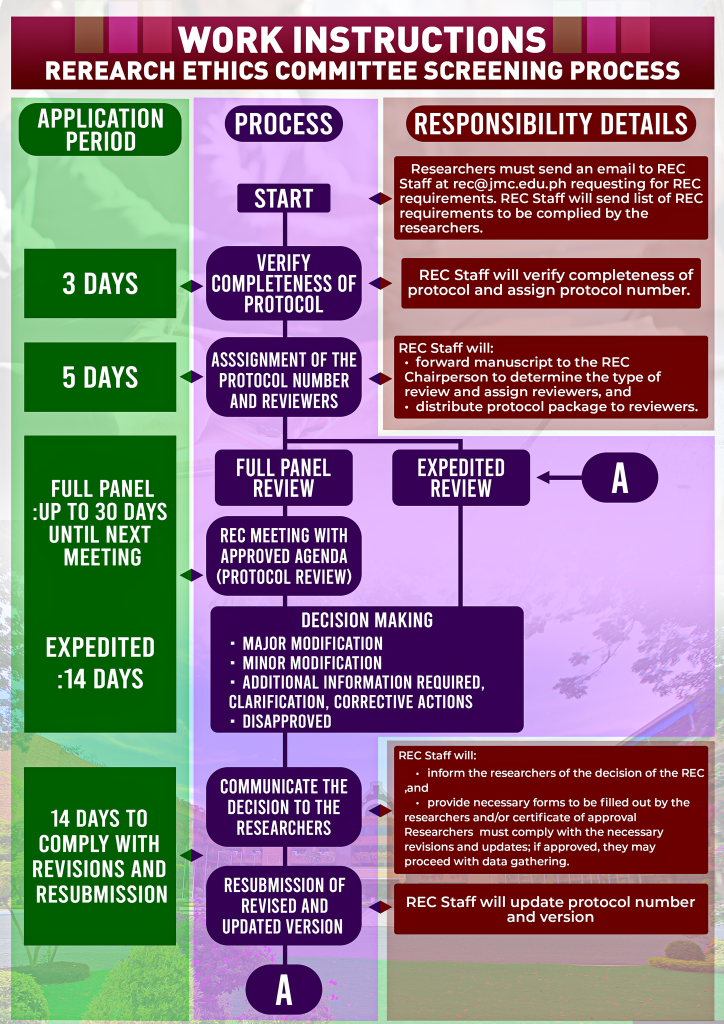
REC Process Flow
This process flow will guide you through the steps and timeline of how your research paper will be reviewed.
Rec List of
Initial Requirements
This list of initial requirements will serve as the foundation for ensuring the protection, dignity, and well-being of all participants involved in the study.
Rec Forms
The following forms outline the initial requirements for submission to the research ethics committee, as well as additional forms required post-approval to maintain compliance and transparency throughout the research process.
Other Pertinent
Documents
These steps outline the responsibilities of the researchers subsequent to the approval of their research conduct.
REC Process Flow
REC List of Requirements

Research Ethics Committee
- Application Form (REC Form 61)
- Letter Request for Review (REC Form 3,6, signed by the adviser/dean)
- Endorsement/Referral Letter (REC Form 3.7, signed by the adviser/dean)
- Full Proposal/Study Protocol (revised manuscript based on your proposal defense
- Certificate of Proposal Defense (c/o Research Dept.)
- Technical Review Approval (Routing Form) – (c/o Research Dept.)
- Curriculum Vitae of Lead Researcher/s
- Confidentiality Agreement (REC Form 41)
- Budget (REC Form 6.13)
- GANTT Chart (REC Form 6.14)
- Dummy Table of Results (REC Form 6.15)
- Informed Consent Form (English, Filipino, or others) (REC Form 5.5) = Assent Form (if applicable – il your participoits are at minor age)
- Receipt of Payment – please proceed to the REC Office in Room 224 for payment. if you are from College of Medicine (COM), disregard this one. This will be part of your miscellaneous fee.
| for undergrad | Php 3,000.0 |
| For College of Medicine (COM) | Php 4,000.0 (covered in Miscellaneous) |
****** Please Note Please designate one representative from your group to handle all communication with the REC. This person will be responsible for emailing requirements, processes, and other protocols-related documents throughout the entire process. This is for REC to easily track protocols-related documents of submissions in the database.
REC FORMS
Form 3.6
Letter Request for Review
Form 3.7
Referral Letter
Form 4.1
Confidentiality Agreement (for JMCFI Members)
Form 5.5 -
Informed Consent Form (ICF)
Form 6.1
Application for Ethics Review of a New Protocol
Form 6.3
Application for Ethics Review of Amendments
Form 6.4
Application for Ethics Review of Progress Report
Form 6.5
Protocol Violation or Deviation Report
Form 6.7
Onsite SAE and SUSAR Reports.docx
Form 6.11
Final Report Form
Form 6.13
Budget
Form 6.14
GANTT Chart
Form 6.15
Dummy Tables
Other Pertinent Documents

Research Ethics Committee
Below are some responsibilities after approval
- Submit Protocol Amendments (REC Form 6.4) (if any) for JMCFI REC approval before implementing them;
- Amendments – refer to any change or revision in the protocol made after its approval from REC
- Submit on-site Serious Adverse Events (SAEs) (REC Form 6.8) and Suspected Unexpected Serious Adverse Reaction (SUSAR) reports (if applicable) to the JMCFI-REC within the day of its detection;
- Submit Progress Report (REC Form 6.5) as determined by the REC based on the level of risk; •
- Submit Final Report (REC Form 6.12) after completion of protocol procedures at the study site;
- Report Protocol Deviations/Violations (REC Form 6.6)
- Protocol Deviation – refers to a non-compliance with the approved protocol that does not increase risk or decrease benefit to participants or does not significantly affect their rights, safety or welfare or the integrity of data.
- Example: a missed visit and any major or minor changes of information after the approval of the protocol are required to be reported in the be reported in the protocol deviation report (change of number of participants or respondents, RRL, research design, research instruments, etc.).
- Protocol Violation – refers to a non-compliance with the approved protocol that increases risk or decreases benefit to participants or significantly affects their rights, safety or welfare or the integrity of data.
- Example: incorrect treatment, non-compliance with inclusion/exclusion criteria.
- Comply with all relevant international and national guidelines and regulations;
- Abide by the principles of Good Research Practice and Ethical Research.
CONNECT with US
Subscribe to our newsletter to hear the latest news



